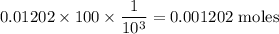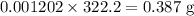Chemistry, 03.11.2020 16:30 elwinelwin9475
The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid must be 0.100 M. Based on this information, what mass of solid conjugate base should the student weigh out to make the buffer solution with a pH of 1.00

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Match the mixture with the substance// i really need on this guys (it’s a pic btw)
Answers: 1

Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
You know the right answer?
The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid m...
Questions

Mathematics, 05.05.2020 10:32



Mathematics, 05.05.2020 10:32


Mathematics, 05.05.2020 10:32



Biology, 05.05.2020 10:32


English, 05.05.2020 10:32



Mathematics, 05.05.2020 10:32






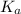 = Acid dissociation constant =
= Acid dissociation constant = 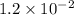
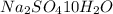 )
)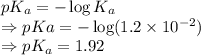
![pH=pK_a+\log\dfrac{[A^-]}{[HA]}\\\Rightarrow pH-pK_a=\log\dfrac{[A^-]}{[HA]}\\\Rightarrow 10^{pH-pK_a}=\dfrac{[A^-]}{[HA]}\\\Rightarrow [A^-]=10^{pH-pK_a}[HA]\\\Rightarrow [A^-]=10^{1-1.92}\times0.1\\\Rightarrow [A^-]=0.01202\ \text{M}](/tpl/images/0863/3238/f5459.png)