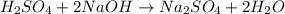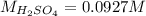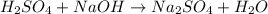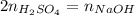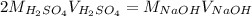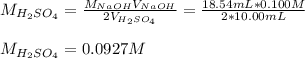Chemistry, 03.11.2020 09:40 kieranoid2017
4) 10.00 mL of sulfuric acid is neutralized in a titration using 18.54 mL of 0.100 M NaOH.
a) Write the neutralization equation for this reaction.
b) What is the concentration of the sulfuric acid?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1


Chemistry, 22.06.2019 20:30
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3
You know the right answer?
4) 10.00 mL of sulfuric acid is neutralized in a titration using 18.54 mL of 0.100 M NaOH.
a) Write...
Questions

Mathematics, 30.03.2021 21:30






Mathematics, 30.03.2021 21:30


Mathematics, 30.03.2021 21:30


Mathematics, 30.03.2021 21:30

Mathematics, 30.03.2021 21:30





Health, 30.03.2021 21:30

Computers and Technology, 30.03.2021 21:30