
A pair of substances undergoes a change. This change is observed in the form of a difference in energy. Substance A falls from energy level M to energy level N and gives off infrared radiation. Substance B also undergoes a change and has energy that is given off and falls from energy level O to energy level P and gives off ultraviolet radiation. Which transition, from M to N or from O to P, has a greater energy difference? Explain your answer. Use a diagram of the electromagnetic spectrum if necessary.
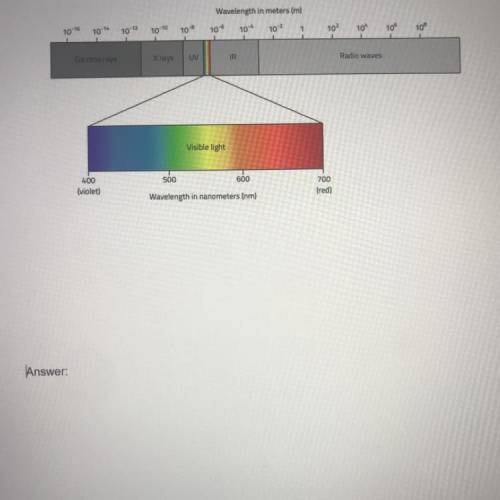

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1


Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3
You know the right answer?
A pair of substances undergoes a change. This change is observed in the form of a difference in ener...
Questions


History, 20.04.2021 01:40

Geography, 20.04.2021 01:40

Computers and Technology, 20.04.2021 01:40

Chemistry, 20.04.2021 01:40

Mathematics, 20.04.2021 01:40

Mathematics, 20.04.2021 01:40


Mathematics, 20.04.2021 01:40


Advanced Placement (AP), 20.04.2021 01:40

Mathematics, 20.04.2021 01:40

Mathematics, 20.04.2021 01:50





Health, 20.04.2021 01:50


History, 20.04.2021 01:50



