
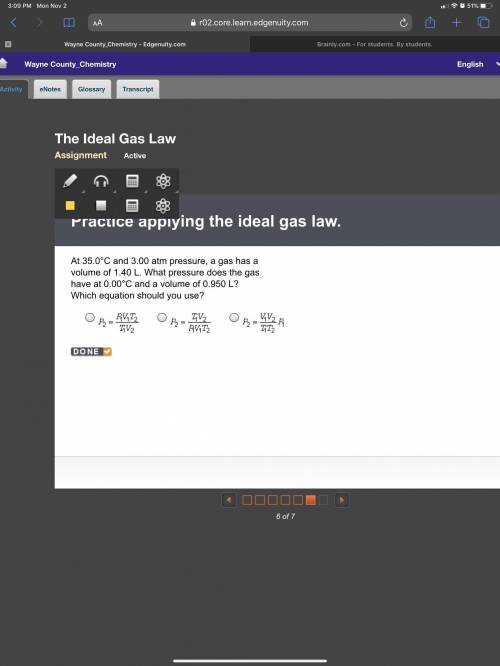
At 35.0°C and 3.00 atm pressure, a gas has a volume of 1.40 L. What pressure does the gas have at 0.00°C and a volume of 0.950 L? Which equation should you use? P subscript 2 equals StartFraction P subscript 1 V subscript 1 T subscript 2 over T subscript 1 V subscript 2 EndFraction. P subscript 2 equals StartFraction T subscript 1 V subscript 2 over P subscript 1 V subscript 1 T subscript 2 EndFraction. P subscript 2 StartFraction equals V subscript 1 V subscript 2 over T subscript 1 T subscript 2 EndFraction P subscript 1.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no]2[o2]
Answers: 3


Chemistry, 22.06.2019 07:30
All cells are made of four types of acids, lipids, proteins, and carbohydrates.
Answers: 1

Chemistry, 22.06.2019 10:00
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1
You know the right answer?
At 35.0°C and 3.00 atm pressure, a gas has a volume of 1.40 L. What pressure does the gas have at 0....
Questions


Mathematics, 10.09.2021 03:20


History, 10.09.2021 03:20

Physics, 10.09.2021 03:20

English, 10.09.2021 03:20

Mathematics, 10.09.2021 03:20

Spanish, 10.09.2021 03:20



Mathematics, 10.09.2021 03:20

Mathematics, 10.09.2021 03:20

Social Studies, 10.09.2021 03:20

History, 10.09.2021 03:20

Mathematics, 10.09.2021 03:20


Mathematics, 10.09.2021 03:20


Mathematics, 10.09.2021 03:20



