Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3



Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3
You know the right answer?
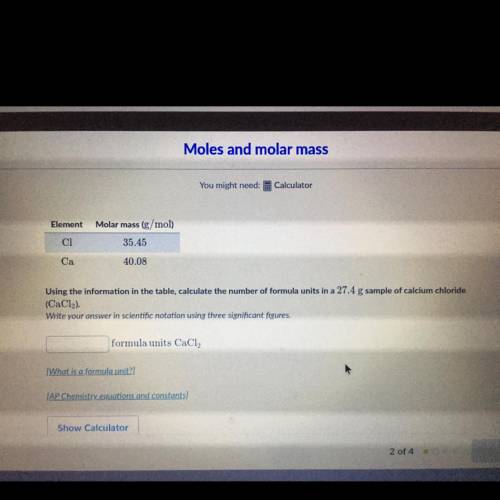
Using the information in the table, calculate the number of formula units in a 27.4 g sample of calc...
Questions


Business, 23.10.2020 15:20






Mathematics, 23.10.2020 15:20

Mathematics, 23.10.2020 15:20






History, 23.10.2020 15:20

English, 23.10.2020 15:20

Chemistry, 23.10.2020 15:20

Mathematics, 23.10.2020 15:20

English, 23.10.2020 15:20