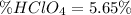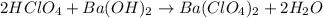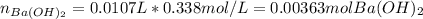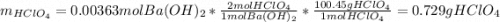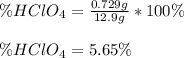Chemistry, 02.11.2020 17:20 sskibi1243
A 12.9 g sample of an aqueous solution of perchloric acid contains an unknown amount of the acid. If 10.7 mL of 0.338 M barium hydroxide are required to neutralize the perchloric acid, what is the percent by mass of perchloric acid in the mixture

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:50
Your roll: experienced electron speech is adressed to: a new "freshman class" of electrons job: write a speech task: you are to pretend that you are giving a speech to a new group of electrons. be sure to mention their placement in an atom, their charge, and their role in chemical bonding (ionic and covalent) be specific!
Answers: 3

Chemistry, 21.06.2019 20:30
The first element on the periodic table of elements is carbon. a. true b. false
Answers: 2

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

You know the right answer?
A 12.9 g sample of an aqueous solution of perchloric acid contains an unknown amount of the acid. If...
Questions

Mathematics, 11.11.2020 23:30





Business, 11.11.2020 23:30

Advanced Placement (AP), 11.11.2020 23:30

Mathematics, 11.11.2020 23:30


Chemistry, 11.11.2020 23:30

Mathematics, 11.11.2020 23:30


Mathematics, 11.11.2020 23:30


SAT, 11.11.2020 23:30

Mathematics, 11.11.2020 23:30

Arts, 11.11.2020 23:30

English, 11.11.2020 23:30