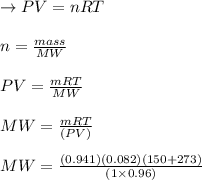A sample of an unknown compound is vaporized at . The gas produced has a volume of at a pressure of , and it weighs . Assuming the gas behaves as an ideal gas under these conditions, calculate the molar mass of the compound. Be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
What does x represent in the formula for the compound xcl4?
Answers: 1

Chemistry, 22.06.2019 08:40
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2

You know the right answer?
A sample of an unknown compound is vaporized at . The gas produced has a volume of at a pressure of...
Questions




Biology, 20.08.2021 01:30


Mathematics, 20.08.2021 01:30

Mathematics, 20.08.2021 01:30




Chemistry, 20.08.2021 01:30





History, 20.08.2021 01:30

Mathematics, 20.08.2021 01:30

Social Studies, 20.08.2021 01:30


Mathematics, 20.08.2021 01:30

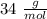 "
"