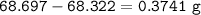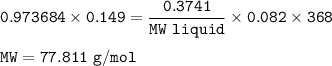Chemistry, 02.11.2020 14:10 joycewingate919
An empty 149 mL flask weighs 68.322 g before a sample of volatile liquid is added. The flask is then placed in a hot (95.0°C) water bath; the barometric pressure is 740 torr. The liquid vaporizes and the gas fills the flask. After cooling, flask and condensed liquid together weigh 68.697 g. What is the molar mass of the liquid?

Answers: 1
Another question on Chemistry


Chemistry, 21.06.2019 18:20
Complete the table for ion charge based upon their losing or gaining electrons in the outer shell. (use the periodic table as necessary.) group most likely ionic charge # of valence electrons i +1 ii +2 iii +3 iv +4 or -4 v -3 vi -2 vii -1 viii 0
Answers: 2


You know the right answer?
An empty 149 mL flask weighs 68.322 g before a sample of volatile liquid is added. The flask is then...
Questions

Social Studies, 02.11.2020 21:20

Mathematics, 02.11.2020 21:20


Mathematics, 02.11.2020 21:20





Biology, 02.11.2020 21:20


Mathematics, 02.11.2020 21:20


Biology, 02.11.2020 21:20

Mathematics, 02.11.2020 21:20

Mathematics, 02.11.2020 21:20

Mathematics, 02.11.2020 21:20

Computers and Technology, 02.11.2020 21:20



Mathematics, 02.11.2020 21:20