The kinetic-molecular theory states that ideal gas molecules do which of
the following?*
are...

Chemistry, 02.11.2020 14:00 katwilf1771
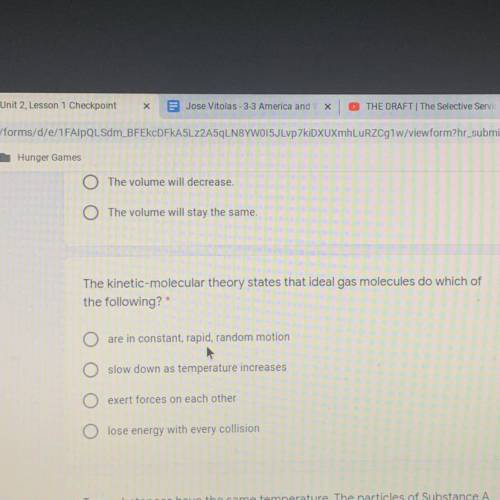
The kinetic-molecular theory states that ideal gas molecules do which of
the following?*
are in constant, rapid, random motion
O slow down as temperature increases
O exert forces on each other
O lose energy with every collision

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Does the temperature affect the solubility of sugar and salt in water? if it does tell me like different temperatures with different solubilities so i can sketch down a graph
Answers: 2

Chemistry, 21.06.2019 20:30
Which of the following true? a_volcanoes and earthquakes often near the plate boundaries. b_volcanoes occur whereve there are tall mountains. c_earthquakes cause volcanoes in the same location to erupt violently d_volcanoes and earthquakes occur only where plates are colliding with each other
Answers: 2


Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1
You know the right answer?
Questions


English, 12.10.2019 10:10


Mathematics, 12.10.2019 10:10







Mathematics, 12.10.2019 10:10

Physics, 12.10.2019 10:10

English, 12.10.2019 10:10


Health, 12.10.2019 10:10







