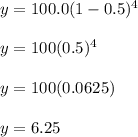Chemistry, 01.11.2020 08:20 cfigueroablan
The half-life of carbon-14 is 5,730 years. If
you started with 100.0 g of carbon-14, how
much would remain after 4 half-lives?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

You know the right answer?
The half-life of carbon-14 is 5,730 years. If
you started with 100.0 g of carbon-14, how
Questions





English, 21.08.2019 20:00






History, 21.08.2019 20:00

Chemistry, 21.08.2019 20:00


History, 21.08.2019 20:00


History, 21.08.2019 20:00

Chemistry, 21.08.2019 20:00


Computers and Technology, 21.08.2019 20:00

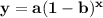 where a is the initial amount of the substance, b is the decay factor, and x is the time elapsed (this is also known as the exponential decay function). We can define our values and substitute them into the equation.
where a is the initial amount of the substance, b is the decay factor, and x is the time elapsed (this is also known as the exponential decay function). We can define our values and substitute them into the equation.