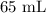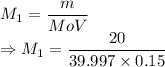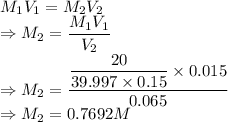Chemistry, 30.10.2020 16:50 kaelah6846
A student prepared a stock solution by dissolving 20.0 g of NaOH in enough water to make 150. mL of solution. She then took 15.0 mL of the stock solution and diluted it with enough water to make 65.0 mL of a final solution. What is the concentration of NaOH for the final solution?
A) O. 411 M
B) 0.534 M
C) 1.87 M
D) 2.43 M

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Why are people not able to scuba dive in the deep part of the ocean
Answers: 2


Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3
You know the right answer?
A student prepared a stock solution by dissolving 20.0 g of NaOH in enough water to make 150. mL of...
Questions

Chemistry, 01.11.2019 04:31




Social Studies, 01.11.2019 04:31

History, 01.11.2019 04:31

Mathematics, 01.11.2019 04:31

English, 01.11.2019 04:31



Geography, 01.11.2019 04:31

History, 01.11.2019 05:31


Mathematics, 01.11.2019 05:31


Mathematics, 01.11.2019 05:31



Mathematics, 01.11.2019 05:31

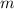 = Mass of sample =
= Mass of sample = 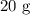
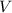 = Volume of solution =
= Volume of solution = 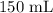
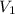 = Initial volume taken out of the stock solution =
= Initial volume taken out of the stock solution = 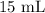
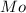 = Molar mass of NaOH =
= Molar mass of NaOH = 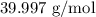
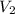 = Final volume of solution =
= Final volume of solution =