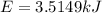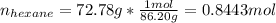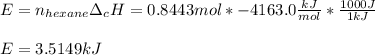The combustion of hexane is given by the following reaction. 2 C 6 H 14 + 19 O 2 → 12 CO 2 + 14 H 2 O The enthalpy of reaction is −4163.0 kJ/mol. How much energy (in joules) will be released if 72.78 grams of hexane is burned. (Molar mass of hexane = 86.20 g/mol).

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 09:10
In a 28 g serving of cheese curls there are 247mg of sodium. how much sodium is in a 12.5 ounce bag
Answers: 1

You know the right answer?
The combustion of hexane is given by the following reaction. 2 C 6 H 14 + 19 O 2 → 12 CO 2 + 14 H 2...
Questions

History, 30.04.2021 16:30



Mathematics, 30.04.2021 16:30


Mathematics, 30.04.2021 16:30

Mathematics, 30.04.2021 16:30


Mathematics, 30.04.2021 16:30


Mathematics, 30.04.2021 16:30

Computers and Technology, 30.04.2021 16:30


Mathematics, 30.04.2021 16:30

Arts, 30.04.2021 16:30



Mathematics, 30.04.2021 16:30