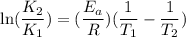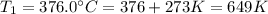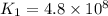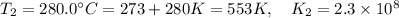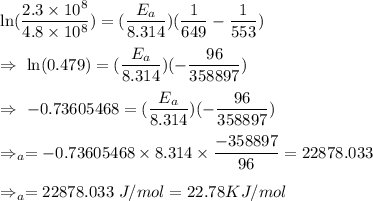The rate constant for a certain reaction is measured at two different temperatures:
Temperature K
376.0°C 4.8 x 10^8
280°C 2.3 x 10^8
Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy for this reaction. Round your answer to significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 23.06.2019 08:10
Time remaining 58: 10 an atom that has 84 protons and 86 neutrons undergoes a reaction. at the end of the reaction, it has 82 protons and 84 neutrons. what happened to the atom? it accepted radiation in a chemical reaction it donated neutrons to another atom in a chemical reaction it emitted an alpha particle in a nuclear reaction. it accepted protons in a nuclear reaction. mark this and retum save and exit next submit
Answers: 3

Chemistry, 23.06.2019 10:30
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1

Chemistry, 23.06.2019 10:30
An atom that gains or loses one or more electrons is called a(n)
Answers: 1
You know the right answer?
The rate constant for a certain reaction is measured at two different temperatures:
Temperature K
Questions

English, 27.07.2021 14:30


English, 27.07.2021 14:30


Social Studies, 27.07.2021 14:30

Chemistry, 27.07.2021 14:30

Physics, 27.07.2021 14:30


Mathematics, 27.07.2021 14:30



Computers and Technology, 27.07.2021 14:30


Computers and Technology, 27.07.2021 14:30

Mathematics, 27.07.2021 14:30

Mathematics, 27.07.2021 14:30

Chemistry, 27.07.2021 14:30

Business, 27.07.2021 14:30

Geography, 27.07.2021 14:30

Social Studies, 27.07.2021 14:30

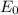 follows by formula:
follows by formula: