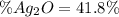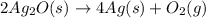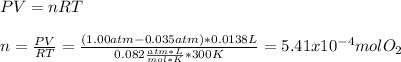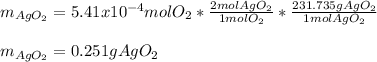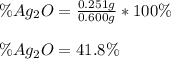A 0.600 g sample containing Ag2O and inert material is heated, causing the silver oxide to decompose according to the following equation: 2 Ag2O(s) → 4 Ag(s) + O2(g) If 13.8 mL of gas are collected over water at 27°C and 1.00 atm external pressure, what is the percentage of silver oxide in the sample? The partial pressure of water is 26.7 mm Hg at 27°C.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which describes interactions between substances and stomata during photosynthesis? check all that apply. oxygen enters stomata. oxygen is released through stomata. carbon dioxide enters stomata. carbon dioxide is released through stomata. hydrogen enters stomata. hydrogen is released through stomata.
Answers: 1

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1

Chemistry, 23.06.2019 03:00
Is it safe to take 450mg of diphenhydramine hydrochloride?
Answers: 1

You know the right answer?
A 0.600 g sample containing Ag2O and inert material is heated, causing the silver oxide to decompose...
Questions

Chemistry, 02.06.2021 16:40

Mathematics, 02.06.2021 16:40

Mathematics, 02.06.2021 16:40

Mathematics, 02.06.2021 16:40


Chemistry, 02.06.2021 16:40

English, 02.06.2021 16:40





History, 02.06.2021 16:40

Mathematics, 02.06.2021 16:40

Mathematics, 02.06.2021 16:40

Mathematics, 02.06.2021 16:40





Mathematics, 02.06.2021 16:40