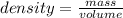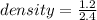Chemistry, 28.10.2020 01:00 blayneaafedt
An object with a mass of 1.2 g takes up a space of 2.4 mL. What is it's density?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3


Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1
You know the right answer?
An object with a mass of 1.2 g takes up a space of 2.4 mL. What is it's density?...
Questions


Advanced Placement (AP), 28.06.2021 07:00

Geography, 28.06.2021 07:00

Mathematics, 28.06.2021 07:00



Business, 28.06.2021 07:00


Mathematics, 28.06.2021 07:00

Mathematics, 28.06.2021 07:00

Mathematics, 28.06.2021 07:00

Geography, 28.06.2021 07:00


Mathematics, 28.06.2021 07:00

English, 28.06.2021 07:00

English, 28.06.2021 07:00



Mathematics, 28.06.2021 07:00

Mathematics, 28.06.2021 07:00