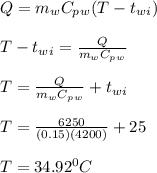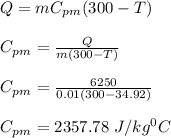Chemistry, 27.10.2020 19:10 Baby010391
A 10.0 g piece of hot metal at 300. °C was dropped into a 150.0 g sample of cooler room temperature water that was initially 25.0 °C. If 6.25 kJ of heat was transferred, what was the final temperature of the water?What was the specific heat of the metal?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1

Chemistry, 23.06.2019 00:20
How many lone pairs of electrons are on the central atom of no3- and what is the molecular shape? one, trigonal planar zero, trigonal pyramidal zero, trigonal planar one, tetrahedral one, trigonal pyramidal
Answers: 1
You know the right answer?
A 10.0 g piece of hot metal at 300. °C was dropped into a 150.0 g sample of cooler room temperature...
Questions


Mathematics, 01.08.2019 08:30

History, 01.08.2019 08:30

Chemistry, 01.08.2019 08:30

Mathematics, 01.08.2019 08:30


Computers and Technology, 01.08.2019 08:30






Mathematics, 01.08.2019 08:30


History, 01.08.2019 08:30

Business, 01.08.2019 08:30


Computers and Technology, 01.08.2019 08:30


Mathematics, 01.08.2019 08:30

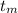 = 300 °C
= 300 °C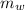 = 150 g = 0.15 kg
= 150 g = 0.15 kg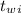 = 25.0 °C
= 25.0 °C