Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2


Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3
You know the right answer?
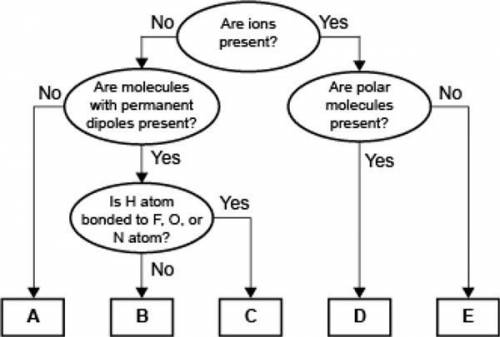
WILL MARK BRAINLIEST. TIMED. PLEASE HELP
A concept map for four types of intermolecular forces and...
Questions


Chemistry, 05.11.2019 09:31

Mathematics, 05.11.2019 09:31

Biology, 05.11.2019 09:31

Mathematics, 05.11.2019 09:31

Physics, 05.11.2019 09:31



Mathematics, 05.11.2019 09:31





History, 05.11.2019 09:31

Mathematics, 05.11.2019 09:31

Mathematics, 05.11.2019 09:31

Mathematics, 05.11.2019 09:31



Mathematics, 05.11.2019 09:31