(GIVING BRAINLIEST)
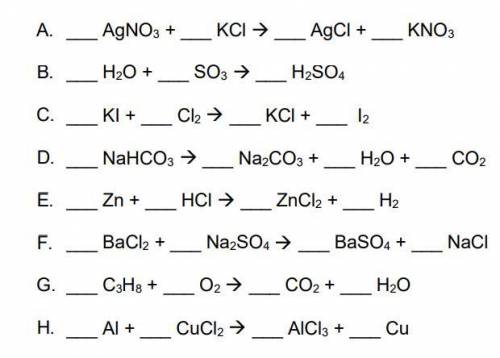
Balance each of the following chemical equations below
...


Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1



You know the right answer?
Questions


Mathematics, 01.09.2020 03:01

Mathematics, 01.09.2020 03:01

Computers and Technology, 01.09.2020 03:01

Advanced Placement (AP), 01.09.2020 03:01






English, 01.09.2020 03:01

Mathematics, 01.09.2020 03:01





Mathematics, 01.09.2020 03:01


Mathematics, 01.09.2020 03:01