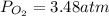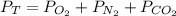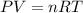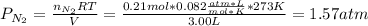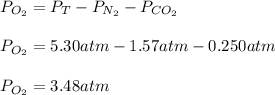Chemistry, 23.10.2020 15:40 kayleahrayne
Suppose that Daniel has a 3.00 L bottle that contains a mixture of O2 , N2 , and CO2 under a total pressure of 5.30 atm. He knows that the mixture contains 0.210 mol N2 and that the partial pressure of CO2 is 0.250 atm. If the temperature is 273 K, what is the partial pressure of O2

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1



Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1
You know the right answer?
Suppose that Daniel has a 3.00 L bottle that contains a mixture of O2 , N2 , and CO2 under a total p...
Questions




English, 19.08.2020 08:01




Mathematics, 19.08.2020 08:01

Mathematics, 19.08.2020 08:01


Biology, 19.08.2020 08:01

Mathematics, 19.08.2020 08:01



Mathematics, 19.08.2020 08:01


Mathematics, 19.08.2020 08:01


Mathematics, 19.08.2020 08:01

Mathematics, 19.08.2020 08:01