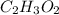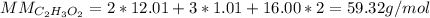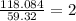Chemistry, 22.10.2020 22:01 tiffanyjadeb
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen.
In an experiment, the molar mass of the compound was determined to be 118.084 g/mol. What is the molecular formula of the compound?
For both questions, show your work or explain how you determined the formulas by giving specific values used in calculations. (10 points)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:10
Which of these will change if the air in aclosed bottle is heated? abcdthe mass of the airthe composition of the airthe air pressure in the bottlethe number of air molecules in the bottle
Answers: 3


Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

You know the right answer?
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen...
Questions




Social Studies, 07.05.2021 04:10





Mathematics, 07.05.2021 04:10






Computers and Technology, 07.05.2021 04:10



Mathematics, 07.05.2021 04:10