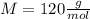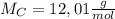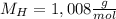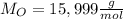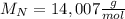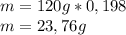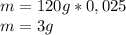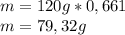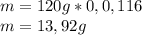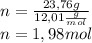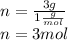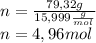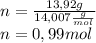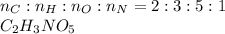Chemistry, 22.10.2020 14:01 pizarroisaid
Peroxyacylnitrate (PAN) is one of the components
of smog. It is a compound of C, H, N, and O.
Determine the percent composition of oxygen and
the empirical formula from the following percent
composition by mass: 19.8 percent C,
2.50 percent
H, 11.6 percent N. What is its molecular formula
given that its molar mass is about 120 g?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 08:30
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3
You know the right answer?
Peroxyacylnitrate (PAN) is one of the components
of smog. It is a compound of C, H, N, and O.
...
...
Questions


Mathematics, 13.11.2020 17:10

Mathematics, 13.11.2020 17:10









History, 13.11.2020 17:10






History, 13.11.2020 17:10