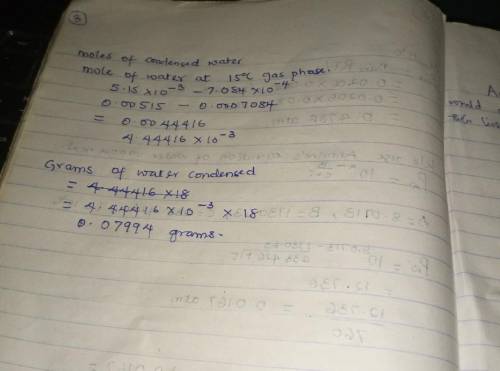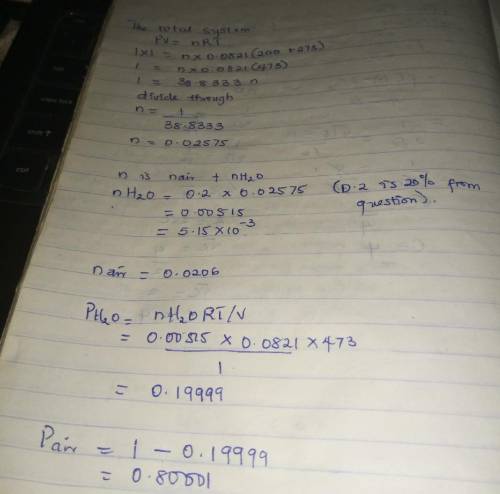Chemistry, 21.10.2020 17:01 smartgirl9989
Air containing 20.0 mol% water vapor at an initial pressure of 1 atm absolute is cooled in a 1- liter sealed vessel from 200°C to 15°C. (a) What is the pressure in the vessel at the end of the process? (Hint: The partial pressure of air in the system can be determined from the expression pair = nairRT/V and P = pair + pH2O. You may neglect the volume of the liquid water condensed, but you must show that condensation occurs.) (b) What is the mole fraction of water in the gas phase at the end of the process? (c) How much water (grams) condenses?

Answers: 2
Another question on Chemistry


Chemistry, 23.06.2019 01:30
Which is an example of a highly unstable isotope that is often used in fission reactions?
Answers: 1

Chemistry, 23.06.2019 02:20
Why dose heating increase the speed at which a solution dissolved in water
Answers: 1

Chemistry, 23.06.2019 12:30
How do you interpret a chromatogram for what mixtures contain?
Answers: 3
You know the right answer?
Air containing 20.0 mol% water vapor at an initial pressure of 1 atm absolute is cooled in a 1- lite...
Questions

Mathematics, 06.10.2019 07:30


Mathematics, 06.10.2019 07:30

Mathematics, 06.10.2019 07:30



English, 06.10.2019 07:30

Mathematics, 06.10.2019 07:30

Biology, 06.10.2019 07:30

Geography, 06.10.2019 07:30

Mathematics, 06.10.2019 07:30

Geography, 06.10.2019 07:30


Mathematics, 06.10.2019 07:30




Geography, 06.10.2019 07:30

Mathematics, 06.10.2019 07:30