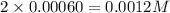Hydrogen sulfide decomposes according to the following reaction, for which Kc=9.30E-8 at 700 degrees Celsius. 2 H2S(g) --> 2 H2(g) + S2(g) If 0.29 moles of H2S is placed in a 3.0-L container, What is the equilibrium concentration of H2(g) at 700 degrees Celsius?

Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 02:00
An alpha particle is: a hydrogen atom a nucleus of helium two neutrons an electron
Answers: 1

Chemistry, 23.06.2019 05:10
Will mark as brainliest ! how many grams of iron metal do you expect to be produced when 245 grams of an 80.5 percent by mass iron (ii) nitrate solution react with excess aluminum metal? show all work needed to solve this problem!
Answers: 1


Chemistry, 23.06.2019 10:00
Which element forms a compound with chlorine with the general formula mci?
Answers: 1
You know the right answer?
Hydrogen sulfide decomposes according to the following reaction, for which Kc=9.30E-8 at 700 degrees...
Questions


Social Studies, 27.01.2020 09:31

Chemistry, 27.01.2020 09:31



Mathematics, 27.01.2020 09:31

History, 27.01.2020 09:31



Biology, 27.01.2020 09:31





Mathematics, 27.01.2020 09:31

World Languages, 27.01.2020 09:31

History, 27.01.2020 09:31

Spanish, 27.01.2020 09:31


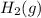 at 700 degrees Celsius is 0.0012 M
at 700 degrees Celsius is 0.0012 M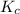
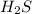 = 0.29 mole
= 0.29 mole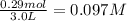
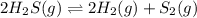
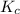 is written as:
is written as:![K_c=\frac{[H_2]^2\times [S_2]}{[H_2S]^2}](/tpl/images/0827/9052/13e21.png)
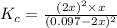
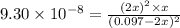
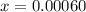
![[H_2]](/tpl/images/0827/9052/08a38.png) = 2x=
= 2x=