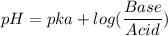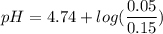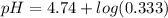Chemistry, 21.10.2020 16:01 anonymous115296
Calculate the expected pH of the buffer after the addition of 1.0 mL of 1M HCl. Remember that you are using 50 mL of the buffer, so be sure to calculate the moles of acetic acid and acetate in 50 mL of the buffer.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2

You know the right answer?
Calculate the expected pH of the buffer after the addition of 1.0 mL of 1M HCl. Remember that you ar...
Questions


Mathematics, 16.01.2020 00:31

Mathematics, 16.01.2020 00:31

Law, 16.01.2020 00:31


Arts, 16.01.2020 00:31


Mathematics, 16.01.2020 00:31


Mathematics, 16.01.2020 00:31




Mathematics, 16.01.2020 00:31


History, 16.01.2020 00:31


English, 16.01.2020 00:31