
Chemistry, 21.10.2020 16:01 ilovecatsomuchlolol
An aqueous solution of Pb(NO3)2 is made by placing 275 g of solid Pb(NO3)2 into a volumetric flask and adding water to the 1.00 L mark (assume that 775g of water has been added to achieve this total solution volume). (Assume MW of Pb(NO3)2 = 331g/mole) A) What is the molarity (M) of this solution? B) What is the molality (m) of this solution? C) What is the mass % of Pb(NO3)2 in this solution? D) What is the mole fraction of Pb(NO3)2 present in this solution?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Aglow stick contains a glass vial with chemicals. when the glow stick is bent, the vial breaks and the chemicals react to produce a glow. a science student observes that a glow stick kept in the freezer glows for a longer duration than a glow stick kept at room temperature. what conclusion can be drawn based on the observation? be sure to note the outcome and test variables in the conclusion.
Answers: 1

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

Chemistry, 23.06.2019 10:30
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 3.75 mol fe and 8.70 mol nio(oh) react?
Answers: 1

Chemistry, 23.06.2019 13:30
The activation energy for a(n) is quite large and usually takes extra energy from the environment, it is normally not a natural spontaneous process. combustion reaction endothermic reaction exothermic reaction catalyzed reaction
Answers: 1
You know the right answer?
An aqueous solution of Pb(NO3)2 is made by placing 275 g of solid Pb(NO3)2 into a volumetric flask a...
Questions

Mathematics, 13.02.2021 20:50

Mathematics, 13.02.2021 20:50

Mathematics, 13.02.2021 20:50

Biology, 13.02.2021 20:50

Mathematics, 13.02.2021 20:50

English, 13.02.2021 20:50

Mathematics, 13.02.2021 20:50

Mathematics, 13.02.2021 20:50


Mathematics, 13.02.2021 20:50






Mathematics, 13.02.2021 20:50

Chemistry, 13.02.2021 20:50


Mathematics, 13.02.2021 20:50

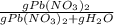 *100%
*100% 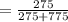 * 100% = 26.2%
* 100% = 26.2%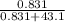 = 0.0189
= 0.0189

