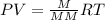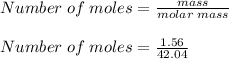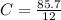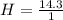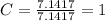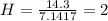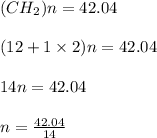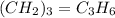Chemistry, 21.10.2020 16:01 charisaramsey
A mixture of cyclopropane gas and oxygen is used as an anesthetic. Cyclopropane contains 85.7% C And 14.3% hydrogen by mass. At 50.0 degrees celcius and .984 atm pressure, 1.56 g cyclopropane has a volume of 1.00L.
Required:
What is the molecular formula of cyclopropane?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 23.06.2019 17:30
Which of the following elements would you expect to have the highest ionization energy value, and why? a. chlorine (cl), because it has a low effective nuclear charge and large radius b. fluorine (f), because it has a large radius and naturally forms a negative ion c. lithium (li), because it has a small radius and naturally forms a positive ion d. neon (ne), because it has a high effective nuclear charge and small radius
Answers: 2

Chemistry, 23.06.2019 18:40
Select the correct answer from each drop-down menu. in a reversible reaction, the forward reaction takes place the reverse reaction. such a reaction, on reaching equilibrium, will have .
Answers: 2
You know the right answer?
A mixture of cyclopropane gas and oxygen is used as an anesthetic. Cyclopropane contains 85.7% C And...
Questions

Mathematics, 28.07.2019 11:40


Biology, 28.07.2019 11:40







Social Studies, 28.07.2019 11:40

Chemistry, 28.07.2019 11:40

Mathematics, 28.07.2019 11:40



Chemistry, 28.07.2019 11:40

Social Studies, 28.07.2019 11:40

Social Studies, 28.07.2019 11:40



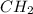 ) is equal to
) is equal to 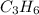
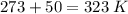 Pressure = 0.984 atm.Mass of cyclopropane = 1.56 grams.Volume of cyclopropane = 1.00 Liter.
Pressure = 0.984 atm.Mass of cyclopropane = 1.56 grams.Volume of cyclopropane = 1.00 Liter.