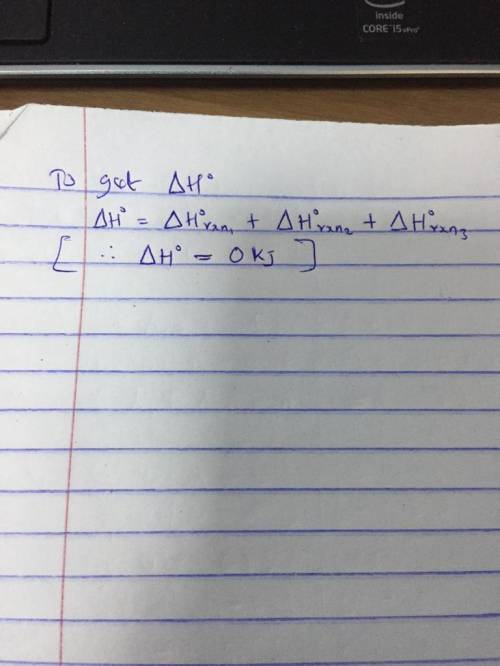6.82
A 2.10-mole sample of crystalline acetic acid, ini-
tially at 17.0°C, is allowed t...

6.82
A 2.10-mole sample of crystalline acetic acid, ini-
tially at 17.0°C, is allowed to melt at 17.0°C and is
then heated to 118.1°C (its normal boiling point) at
1.00 atm. The sample is allowed to vaporize at.
118.1°C and is then rapidly quenched to 17.0°C, so
that it recrystallizes. Calculate AH° for the total pro-
cess as described.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Which type of stress results when two plates push against one another? a. compression b. tension c. force d. shear
Answers: 1


Chemistry, 22.06.2019 22:00
11) burning your hand when accidentally touching a hot plate is an example of which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 2

Chemistry, 23.06.2019 05:40
Why is it incorrect to balance a chemical equation by changing the subscripts? explain.
Answers: 2
You know the right answer?
Questions

Mathematics, 15.04.2021 23:50



Health, 15.04.2021 23:50

Mathematics, 15.04.2021 23:50





Business, 15.04.2021 23:50

Chemistry, 15.04.2021 23:50



Mathematics, 15.04.2021 23:50


Mathematics, 15.04.2021 23:50


Mathematics, 15.04.2021 23:50

Mathematics, 15.04.2021 23:50

Mathematics, 15.04.2021 23:50