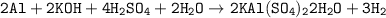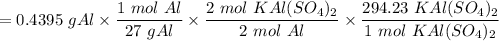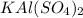Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:40
If 10.0 ml of the solution on the right are withdrawn from the 100 ml beaker and diluted again in a similar manner, what is the new concentration? m nacl
Answers: 2


Chemistry, 22.06.2019 01:30
Sulfuric acid (a component of acid rain) reacts with limestone (calcium carbonate) to produce calcium sulfate and carbon dioxide. this damages buildings and statues made of limestone. which solution of sulfuric acid will damage these structures more quickly? a. 0.001% b. 0.005% c. 0.010% d. 0.015%
Answers: 3

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3
You know the right answer?
g A 0.4395 g sample of aluminum reacts according to our experiment to produce alum. 5.1629 g of drie...
Questions

History, 04.03.2021 01:40




Mathematics, 04.03.2021 01:40


Biology, 04.03.2021 01:40

Mathematics, 04.03.2021 01:40

Mathematics, 04.03.2021 01:40

English, 04.03.2021 01:40

English, 04.03.2021 01:40

Mathematics, 04.03.2021 01:40


Computers and Technology, 04.03.2021 01:40





Mathematics, 04.03.2021 01:40

Biology, 04.03.2021 01:40