
Chemistry, 20.10.2020 04:01 tucecoskun
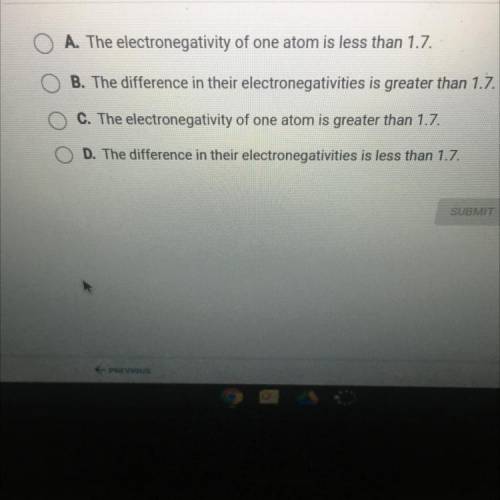
Which sentence describes two atoms that form a covalent bond?
A. The electronegativity of one atom is less than 1.7
B. The difference in their electronegativites is greater than 1.7
C. The electronegativity of one atom is greater than 1.7
D. The difference in their electronegativities is less than 1.7

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 12:30
The electron configurations of two different atoms are shown below. each yellow electron has a charge of 1−, and the net charge of each nucleus is shown. these atoms will combine with bond. a. an ionic b. a positive c. a negative d. a covalent plzzz mee with !
Answers: 1

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 1
You know the right answer?
Which sentence describes two atoms that form a covalent bond?
A. The electronegativity of one atom...
Questions


Mathematics, 12.07.2019 23:00





History, 12.07.2019 23:00

Mathematics, 12.07.2019 23:00

Chemistry, 12.07.2019 23:00

English, 12.07.2019 23:00

Mathematics, 12.07.2019 23:00

History, 12.07.2019 23:00

Mathematics, 12.07.2019 23:00

Mathematics, 12.07.2019 23:00


Geography, 12.07.2019 23:00

Geography, 12.07.2019 23:00

Geography, 12.07.2019 23:00


World Languages, 12.07.2019 23:00



