
Chemistry, 19.10.2020 22:01 montgomerykarloxc24x
When nitrogen, oxygen, fluorine, sodium, magnesium and aluminum ionize, they all will have:
a. different electron configuration from each other.
b. an unchanged electron configuration.
c. the same charge.
d. the same electron configuration (isoelectronic) as neon.
[Definition: The word isoelectronic means that when you write out the electron configuration they are the same. An exam would be He and Li whereby both of them have 2 electrons and therefore they are both are 1s2 in their electron configurations.]

Answers: 2
Another question on Chemistry


Chemistry, 23.06.2019 04:31
One student said that the investigation was not valid (a fair test). write a plan for the investigation that includes improvements to the method and apparatus
Answers: 1

Chemistry, 23.06.2019 11:30
If a refrigerator is a heat pump that follows the first law of thermodynamics, how much heat was removed from food inside of the refrigerator if it released 300j of energy to the room?unit:
Answers: 1

Chemistry, 23.06.2019 17:00
What statement about the energy of a phase change is true
Answers: 1
You know the right answer?
When nitrogen, oxygen, fluorine, sodium, magnesium and aluminum ionize, they all will have:
a. diff...
Questions

Law, 21.09.2019 19:30

Biology, 21.09.2019 19:30


Mathematics, 21.09.2019 19:30



English, 21.09.2019 19:30


History, 21.09.2019 19:30


Computers and Technology, 21.09.2019 19:30



Biology, 21.09.2019 19:30


History, 21.09.2019 19:30

Computers and Technology, 21.09.2019 19:30

English, 21.09.2019 19:30


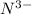 will have electronic configuration of 2,8 ( same as that of neon)
will have electronic configuration of 2,8 ( same as that of neon)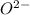 will have electronic configuration of 2,8 ( same as that of neon)
will have electronic configuration of 2,8 ( same as that of neon)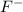 will have electronic configuration of 2,8 ( same as that of neon)
will have electronic configuration of 2,8 ( same as that of neon)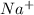 will have electronic configuration of 2,8 ( same as that of neon)
will have electronic configuration of 2,8 ( same as that of neon)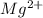 will have electronic configuration of 2,8 ( same as that of neon)
will have electronic configuration of 2,8 ( same as that of neon)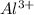 will have electronic configuration of 2,8 ( same as that of neon)
will have electronic configuration of 2,8 ( same as that of neon)

