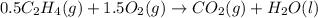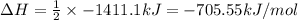Chemistry, 19.10.2020 14:01 samyajones68
Using the reaction below: 2 CO2(g) + 2 H2O(l) → C2H4(g) + 3 O2(g) ΔHrxn= +1411.1 kJ What would be the heat of reaction for this reaction? 0.5 C2H4(g) + 1.5 O2(g) → CO2(g) + H2O(l) ΔHrxn= ??? KJ Question 6 options: a) Not enough information is given b) -2822.2 kJ c) +1411.1 kJ d) -705.55 kJ e) -1411.1 kJ

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 22.06.2019 16:30
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1

You know the right answer?
Using the reaction below: 2 CO2(g) + 2 H2O(l) → C2H4(g) + 3 O2(g) ΔHrxn= +1411.1 kJ What would be th...
Questions



Mathematics, 30.06.2019 01:00

Mathematics, 30.06.2019 01:00

Mathematics, 30.06.2019 01:00


Computers and Technology, 30.06.2019 01:00

History, 30.06.2019 01:00


Computers and Technology, 30.06.2019 01:00

Chemistry, 30.06.2019 01:00

Computers and Technology, 30.06.2019 01:00

Mathematics, 30.06.2019 01:00

Health, 30.06.2019 01:00

Mathematics, 30.06.2019 01:00

History, 30.06.2019 01:00

Mathematics, 30.06.2019 01:00


Mathematics, 30.06.2019 01:00

English, 30.06.2019 01:00

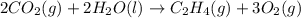
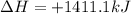
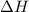
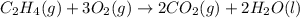
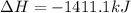
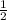 , enthalpy gets half:
, enthalpy gets half: