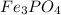Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

You know the right answer?
A 13.58 g sample of a compound contains 8.67 g of iron, Fe, 1.60 g of phosphorus, P, and oxygen, O....
Questions


Health, 27.08.2019 06:20

Mathematics, 27.08.2019 06:20

Health, 27.08.2019 06:20

History, 27.08.2019 06:20

English, 27.08.2019 06:20

English, 27.08.2019 06:20

World Languages, 27.08.2019 06:20

Mathematics, 27.08.2019 06:20



Physics, 27.08.2019 06:20

Chemistry, 27.08.2019 06:20



Mathematics, 27.08.2019 06:20


Chemistry, 27.08.2019 06:20