
Chemistry, 18.10.2020 14:01 tinyturtles
Aluminum metal reacts with chlorine gas to form aluminum chloride. The reaction can be represented by the following balanced chemical equation:
2 Al(s) + 3 Cl2(g) → 2 AlCl3(s).
If you were trying to react 52.9 grams of chlorine gas completely, how many grams of aluminum would you need?
[grams of Aluminum] Do NOT enter the unit in your answer. Report your answer with 3 SFs.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1

Chemistry, 22.06.2019 22:30
What is the work done by the electric force to move a 1 c charge from a to b?
Answers: 2
You know the right answer?
Aluminum metal reacts with chlorine gas to form aluminum chloride. The reaction can be represented b...
Questions




History, 04.08.2019 09:30

Geography, 04.08.2019 09:30

Biology, 04.08.2019 09:30

Mathematics, 04.08.2019 09:30


Advanced Placement (AP), 04.08.2019 09:30


Mathematics, 04.08.2019 09:30



Mathematics, 04.08.2019 09:30


History, 04.08.2019 09:30


History, 04.08.2019 09:30

History, 04.08.2019 09:30

History, 04.08.2019 09:30

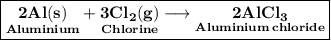
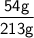 of Aluminium.
of Aluminium.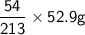 =
=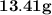 of Aluminium.
of Aluminium.

