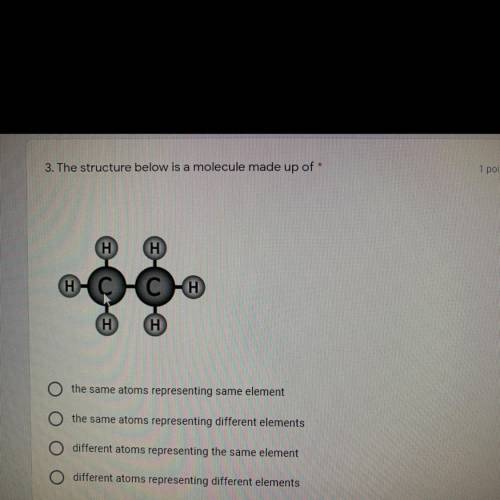
This is for chemistry:/
...

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 03:30
Explain why pure hydrogen cyanide does not conduct electricity, but become a conductor when it is dissolved in water? (at room temp, pure hcn exists as a volatile liquid)
Answers: 1

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3

You know the right answer?
Questions

Mathematics, 18.11.2020 14:00

English, 18.11.2020 14:00

Geography, 18.11.2020 14:00

Mathematics, 18.11.2020 14:00

Computers and Technology, 18.11.2020 14:00


Mathematics, 18.11.2020 14:00



Mathematics, 18.11.2020 14:00



Biology, 18.11.2020 14:00

Medicine, 18.11.2020 14:00

Social Studies, 18.11.2020 14:00


Biology, 18.11.2020 14:00

Physics, 18.11.2020 14:00

Mathematics, 18.11.2020 14:00