Chemistry, 17.10.2020 20:01 ahmedeldyame
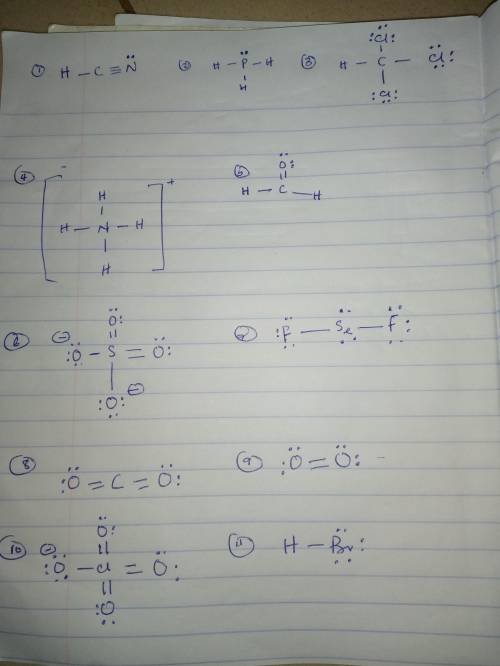
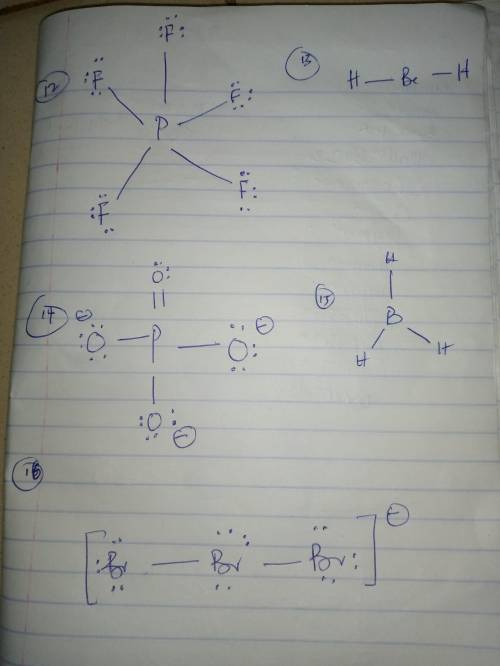
For each of the following molecules draw the Lewis structure on a separate sheet of paper. MAKE SURE TO FOLLOW THE RULES FROM CLASS (ie do not break the octet rule unless necessary to connect all the atoms). Then based on your structure indicate:
the total number of valence electrons.
the electronic and molecular shapes (choose from: linear, trigonal planar, bent, tetrahedral, trigonal pyramidal, trigonal bipyramidal, seesaw, T-shaped, octahedral, square pyramidal, or square planar).
whether or not the molecule is polar (Y/N).
Note: The central atom is the first atom listed, except for HCN, H2CO, and OCN-, where carbon is the central atom (underlined).
FormulaValence electronsElectronic ShapeMolecular ShapePolar (Y/N)
HCN
PH3
CHCl3
NH4+
H2CO
SO42-
SeF2
CO2
O2
ClO4-
HBr
PF5
BeH2
PO43-
BH3
Br3-

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1

Chemistry, 22.06.2019 12:00
Most materials are not magnetic because their magnetism has worn off. their magnetic domains are arranged randomly. they lack magnetic fields. earth’s heat has destroyed their magnetism.
Answers: 1

Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1
You know the right answer?
For each of the following molecules draw the Lewis structure on a separate sheet of paper. MAKE SURE...
Questions

Mathematics, 25.11.2019 04:31


Mathematics, 25.11.2019 04:31


Advanced Placement (AP), 25.11.2019 04:31

Mathematics, 25.11.2019 04:31

English, 25.11.2019 04:31

Biology, 25.11.2019 04:31

Spanish, 25.11.2019 04:31

Mathematics, 25.11.2019 04:31

Mathematics, 25.11.2019 04:31


English, 25.11.2019 04:31

Computers and Technology, 25.11.2019 04:31


Mathematics, 25.11.2019 04:31


History, 25.11.2019 04:31

Social Studies, 25.11.2019 04:31