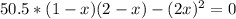Chemistry, 16.10.2020 14:01 williamsjako
A 1.00-L flask is filled with 1.00 moles of H2 and 2.00 moles of I2. The value of the equilibrium constant for the reaction of hydrogen and iodine reacting to form hydrogen iodide is 50.5 under the given conditions. What are the equilibrium concentrations of H2 , I2 , and HI in moles/L? H2 (g) + I2 (g) ⇌ 2HI(g)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:00
The overall chemical reaction for photosynthesis isshown below: 6co2 + 6h20 → c6h12o6 + 602what mass of glucose (c6h1206) can form from71.89 g co2? (molar mass of c6h1206 = 180.18g/mol; molar mass of co2 = 44.01 g/mol)71.89 g co2=g c6h1206
Answers: 1

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1
You know the right answer?
A 1.00-L flask is filled with 1.00 moles of H2 and 2.00 moles of I2. The value of the equilibrium co...
Questions







Mathematics, 18.04.2020 07:24




Social Studies, 18.04.2020 07:24

Mathematics, 18.04.2020 07:24

Chemistry, 18.04.2020 07:24



Social Studies, 18.04.2020 07:25


Physics, 18.04.2020 07:25

SAT, 18.04.2020 07:25

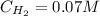
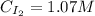
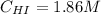
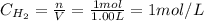
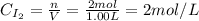
![Kc = \frac{[HI]^{2}}{[H_{2}][I_{2}]} = \frac{(2x)^{2}}{(1-x)(2-x)}](/tpl/images/0811/0470/b43fa.png)