
Chemistry, 16.10.2020 09:01 suttonfae336
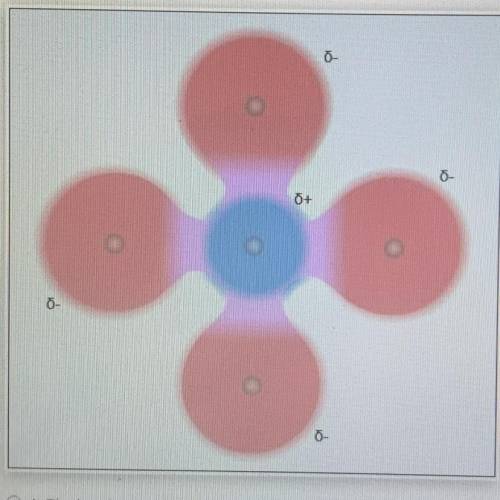
3. Which of the following statements is true about the molecule shown below?
O A. The bonds are polar and the molecule is polar.
O B. The bonds are polar and the molecule is nonpolar.
O C. The bonds are nonpolar and the molecule is polar
O D. The bonds are nonpolar and the molecule is nonpolar

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 23.06.2019 03:00
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 1
You know the right answer?
3. Which of the following statements is true about the molecule shown below?
O A. The bonds are pol...
Questions

Mathematics, 03.03.2021 18:10


Mathematics, 03.03.2021 18:10

Social Studies, 03.03.2021 18:10

Chemistry, 03.03.2021 18:10

Social Studies, 03.03.2021 18:10

Mathematics, 03.03.2021 18:10


Mathematics, 03.03.2021 18:10



Health, 03.03.2021 18:10

Mathematics, 03.03.2021 18:10


Mathematics, 03.03.2021 18:10



Mathematics, 03.03.2021 18:10

Social Studies, 03.03.2021 18:10



