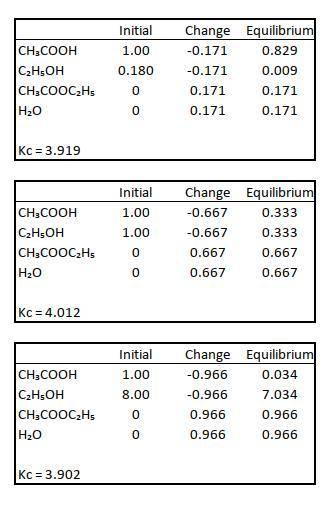
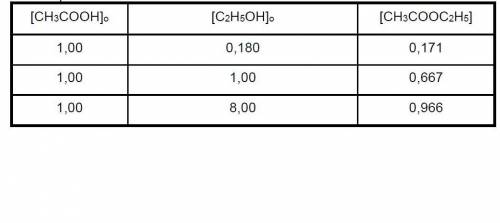
Data for CH3COOH(l) + C2H5OH(l) CH3COOC2H5(l) + H2O(l) balance were obtained at 100. The initial concentrations of the reagents are indicated in columns 1 and 2 of the table below and the CH3COOC2H5 concentrations in equilibrium are given in column 3. Calculate H2O and determine the value of KC

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1

Chemistry, 21.06.2019 20:30
9. write the chemical equation for the following word equations. include symbols for physical states in the equation. a. solid zinc sulfide + oxygen gas -> solid zinc oxide + sulfur dioxide gas b. aqueous hydrochloric acid + aqueous barium hydroxide -> aqueous barium chloride + water
Answers: 1


Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
You know the right answer?
Data for CH3COOH(l) + C2H5OH(l) CH3COOC2H5(l) + H2O(l) balance were obtained at 100. The initial con...
Questions

History, 25.02.2021 01:10




Social Studies, 25.02.2021 01:10




Chemistry, 25.02.2021 01:10





Mathematics, 25.02.2021 01:10

Mathematics, 25.02.2021 01:10

History, 25.02.2021 01:10

Computers and Technology, 25.02.2021 01:10

Mathematics, 25.02.2021 01:10

Biology, 25.02.2021 01:10