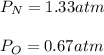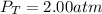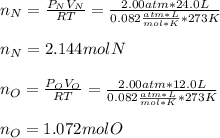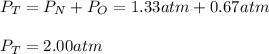In a chemistry laboratory, a student filled a 10.0 L container with two (2) different gases. The
gases are nitrogen gas taken from 24.0 L container at 2.00 atm and 12.0 L container of
oxygen at 2.00 atm. If the temperature of the gases is 273 K, calculate the partial pressure
of both gases in the resulting mixture and the total pressure.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Using the periodic table, complete the table to describe each atom. type in your answers.a ? b? c? d? e? f?
Answers: 1

Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2


Chemistry, 23.06.2019 13:30
32p and 31p are two isotopes of phosphorus. compare the number if subatomic particles that are present in the atoms of these isotopes.
Answers: 1
You know the right answer?
In a chemistry laboratory, a student filled a 10.0 L container with two (2) different gases. The
ga...
Questions



Mathematics, 30.08.2019 05:50




History, 30.08.2019 05:50


Geography, 30.08.2019 05:50

Mathematics, 30.08.2019 05:50

English, 30.08.2019 05:50

History, 30.08.2019 05:50



Health, 30.08.2019 05:50




Social Studies, 30.08.2019 05:50