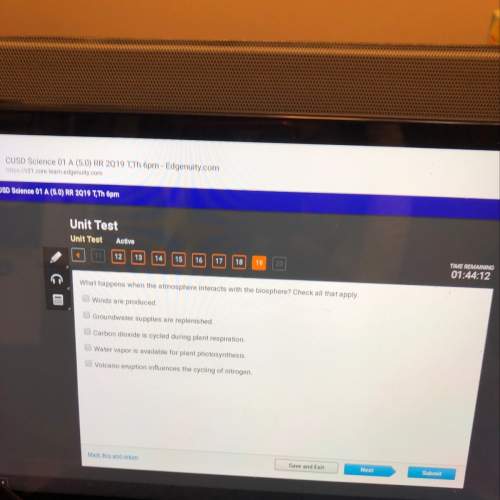
Use the standard heats of formation provided to choose the choice that show the valid math statement and math claim to calculate the standard heat of reaction for the reaction between hydrogen gas and oxygen gas shown.
2H2 (g) + O2 (g) → 2H2O (l)
Substance ΔH°f (kJ/mol)
H2 (g) 0
O2 (g) 0
H20 (l) -285.8
A)(0-0) - (-285) = 285 kJ/mol
B)0 - (-285-0) = -285 kJ/mol
C)-285 - (0-0) = -285 kJ/mol
D)(-285-0) - (0) = 285 kJ/mol

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which actions would increase the rate at salt dissolves in water? stir the water? crush the salt? use less water? heat the water? cool the salt
Answers: 3



Chemistry, 22.06.2019 12:00
An atom of which element reacts with an atom of hydrogen to form a bond with the greatest degree of polarity ?
Answers: 1
You know the right answer?
Use the standard heats of formation provided to choose the choice that show the valid math statement...
Questions


Physics, 12.07.2019 23:30


Physics, 12.07.2019 23:30

Mathematics, 12.07.2019 23:30

History, 12.07.2019 23:30

History, 12.07.2019 23:30

History, 12.07.2019 23:30




Mathematics, 12.07.2019 23:30


Spanish, 12.07.2019 23:30





Computers and Technology, 12.07.2019 23:30