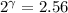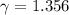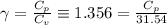The constant volume heat capacity of a gas can be measured by observing the decrease in temperature when it expands adiabatically and reversibly. If the decrease in pressure is also measured, we can use it to infer the value of γ = Cp/Cv and hence, by combining the two values, deduce the constant-pressure heat capacity. A fluorocarbon gas was allowed to expand reversibly and adiabatically to twice its volume; as a result, the temperature fell from 298.15 K to 248.44 K and its pressure fell from 202.94 kPa to 81.840 kPa. Evaluate Cp

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
According to periodic trend, which of the following most likely has the highest ionization energy? kr be ni sc
Answers: 3


Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 15:30
Which suspect most likely committed the robbery and how do you know
Answers: 2
You know the right answer?
The constant volume heat capacity of a gas can be measured by observing the decrease in temperature...
Questions


Mathematics, 25.06.2021 04:20

Arts, 25.06.2021 04:20

Chemistry, 25.06.2021 04:20






Geography, 25.06.2021 04:20




Mathematics, 25.06.2021 04:20

Mathematics, 25.06.2021 04:20

Mathematics, 25.06.2021 04:20




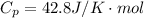
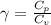
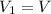

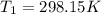
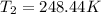
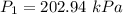
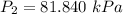
![T_2 = T_1 * [ \frac{V_1}{V_2} ]^{\frac{R}{C_v} }](/tpl/images/0802/4357/d7f78.png)
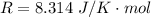
![248.44 = 298.15 * [ \frac{V}{2V} ]^{\frac{8.314}{C_v} }](/tpl/images/0802/4357/a72d7.png)
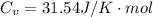
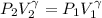
![[ 81.840 *10^3] [2V]^{\gamma} = [202.94 *10^3] V^{\gamma}](/tpl/images/0802/4357/43b7e.png)