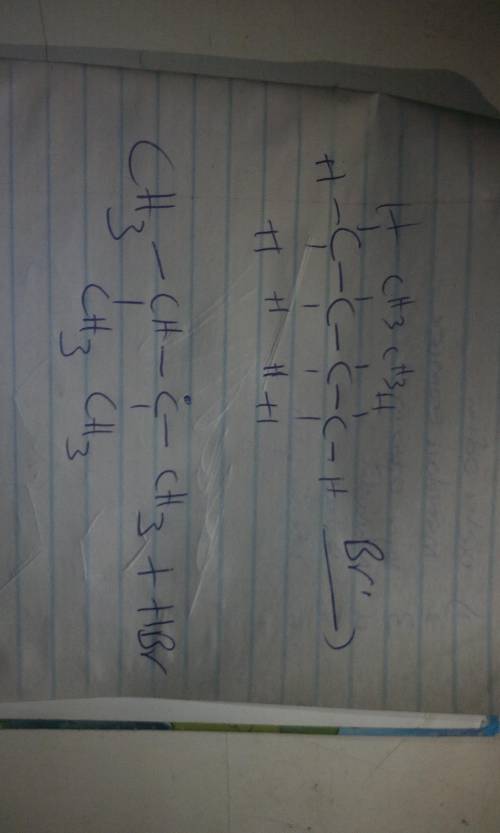In the first propagation step of the bromination of 2,3-dimethylbutane, the newly formed Br radicals react with the molecules of 2,3-dimethylbutane. Give the major products formed by the first propagation step including both organic and inorganic products. Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. Include all free radicals by right-clicking on an atom on the canvas and then using the Atom properties to select the monovalent radical.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 22.06.2019 21:30
An atomic nucleus is composed ofa)protons.b)protons and neutrons.c)protons and electrons.d)protons, neutrons, and electrons.
Answers: 1

Chemistry, 23.06.2019 10:30
Identify the limiting reactant when 9.65-g h2so4 reacts with 6.10-g of naoh.the equation is h2s04 + 2naoh = 2h2o + na2so4• what is the theoretical yield of na2so4, in grams? • how much of the excess reagent will remain after the reaction has been completed? • if 10.5-g of na2so4 are actually recovered experimentally, what is the percent yield?
Answers: 3

Chemistry, 23.06.2019 11:30
How do you calculate the mass of a product when the amounts of more than one reactant are given?
Answers: 3
You know the right answer?
In the first propagation step of the bromination of 2,3-dimethylbutane, the newly formed Br radicals...
Questions




Social Studies, 05.05.2020 03:10




Mathematics, 05.05.2020 03:10

Mathematics, 05.05.2020 03:10


Biology, 05.05.2020 03:10

History, 05.05.2020 03:10

Mathematics, 05.05.2020 03:10

Mathematics, 05.05.2020 03:10

Mathematics, 05.05.2020 03:10


Mathematics, 05.05.2020 03:10