Chemistry, 08.10.2020 07:01 musiclyhollywoodbabo
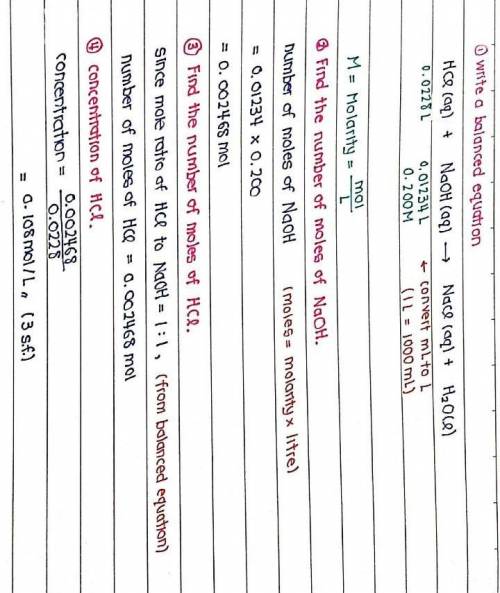
The titration of 22.80 mL of HCl solution of unknown concentration requires 12.34 mL of a 0.200 M NaOH solution to reach the equivalence point. What is the concentration of the unknown HCl solution in M? Express your answer in moles per liter to three significant figures.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 10:50
Determine the empirical formula for succinic acid that is composed of 40.60% carbon, 5.18% hydrogen, and 54.22% oxygen.
Answers: 1
You know the right answer?
The titration of 22.80 mL of HCl solution of unknown concentration requires 12.34 mL of a 0.200 M Na...
Questions

Mathematics, 22.09.2019 20:30

Mathematics, 22.09.2019 20:30

History, 22.09.2019 20:30

English, 22.09.2019 20:30


Physics, 22.09.2019 20:30

Mathematics, 22.09.2019 20:30


Business, 22.09.2019 20:30

Mathematics, 22.09.2019 20:30



Business, 22.09.2019 20:30

Mathematics, 22.09.2019 20:30


History, 22.09.2019 20:30

Mathematics, 22.09.2019 20:30


History, 22.09.2019 20:30