
How many σ and π bonds are in this molecule? A chain of five carbon atoms. There is a double bond between the first and second carbon atoms and a triple bond between the fourth the fifth carbon atoms. There are single bonds between the remaining carbon atoms. There are two hydrogen atoms bonded to the first carbon atom through single bonds, and a single hydrogen atom bonded to both the second and fifth carbon atoms through single bonds. There is an oxygen atom bonded to the third carbon atom through a double bond.

Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 02:50
What is the typical rotational frequency frot for a molecule like n2 at room temperature (25∘c)? assume that d for this molecule is 1å=10−10m. take the total mass of an n2 molecule to be mn2=4.65×10−26kg. you will need to account for rotations around two axes (not just one) to find the correct frequency. express frot numerically in hertz, to three significant figures.
Answers: 3

Chemistry, 23.06.2019 06:30
How can the number of core electrons be determined from the periodic table
Answers: 1

Chemistry, 23.06.2019 11:30
Which part of the healthcare system could best explain how a pharmaceutical drug works
Answers: 2

Chemistry, 23.06.2019 11:40
Which of the following would have the lowest average kinetic energy
Answers: 1
You know the right answer?
How many σ and π bonds are in this molecule? A chain of five carbon atoms. There is a double bond be...
Questions



Mathematics, 12.05.2021 21:20

English, 12.05.2021 21:20



English, 12.05.2021 21:20



Mathematics, 12.05.2021 21:20


Geography, 12.05.2021 21:20


Mathematics, 12.05.2021 21:20

Mathematics, 12.05.2021 21:20

Mathematics, 12.05.2021 21:20

English, 12.05.2021 21:20

English, 12.05.2021 21:20

Mathematics, 12.05.2021 21:20

Mathematics, 12.05.2021 21:20

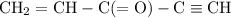 .
.

