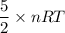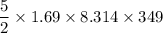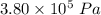A tank contains 120.0 g chlorine gas (Cl2), which is at temperature 76.0°C and absolute pressure 5.70 ✕ 105 Pa. The temperature of the air outside the tank is 19.0°C. The molar mass of Cl2 is 70.9 g/mol. (a) What is the volume of the tank (in m3)? m3 (b) What is the internal energy of the gas (in J)? J (c) What is the work done by the gas (in J) if the temperature and pressure inside the tank drop to 31.0°C and 3.80 ✕ 105 Pa, respectively, due to a leak? (Assume that the air outside the tank can be treated as a vacuum.)

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1

Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2

Chemistry, 22.06.2019 19:50
Which sentence from holes contains an implied personality trait? stanley and his parents had tried to pretend that he was just going away to camp for a while, just like rich kids do. he'd just been in the wrong place at the wrong time. stanley felt somewhat dazed as the guard unlocked his handcuffs and led him off the bus. stanley nodded to show he understood
Answers: 3
You know the right answer?
A tank contains 120.0 g chlorine gas (Cl2), which is at temperature 76.0°C and absolute pressure 5.7...
Questions



English, 17.10.2020 07:01

Computers and Technology, 17.10.2020 07:01



Mathematics, 17.10.2020 07:01

History, 17.10.2020 07:01


Mathematics, 17.10.2020 07:01





Mathematics, 17.10.2020 07:01


Mathematics, 17.10.2020 07:01


Mathematics, 17.10.2020 07:01

English, 17.10.2020 07:01

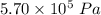
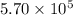
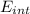 =
=