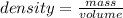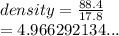Chemistry, 06.10.2020 14:01 lovvyDovvy04
An unknown sample is put in a large graduated cylinder. The water
level in the graduated cylinder was 20.5 mL and the final level
was 38.3 mL. If the mass of the object was determined to be
88.4 g, what is the density of the metal?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3


Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3
You know the right answer?
An unknown sample is put in a large graduated cylinder. The water
level in the graduated cylinder w...
Questions

English, 21.09.2020 18:01

Mathematics, 21.09.2020 18:01





History, 21.09.2020 18:01

English, 21.09.2020 18:01

Geography, 21.09.2020 18:01







Mathematics, 21.09.2020 18:01

Mathematics, 21.09.2020 18:01

Mathematics, 21.09.2020 18:01


Biology, 21.09.2020 18:01