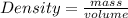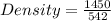Chemistry, 05.10.2020 22:01 LeoInc6806
A cube of metal weighs 1.45 kg and displaces 542 mL of water when immersed. Calculate the density of the metal.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 23.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3

Chemistry, 23.06.2019 06:30
The velocity of any object depends upon a) the location of the object. b) the location of the observer. c) which measurement tools are used. d) the relative motion of the observer.
Answers: 1
You know the right answer?
A cube of metal weighs 1.45 kg and displaces 542 mL of water when immersed. Calculate the density of...
Questions

Mathematics, 30.01.2021 04:50

Chemistry, 30.01.2021 04:50



Mathematics, 30.01.2021 04:50


Chemistry, 30.01.2021 04:50


Chemistry, 30.01.2021 04:50


Mathematics, 30.01.2021 04:50

Mathematics, 30.01.2021 04:50


Mathematics, 30.01.2021 04:50

Mathematics, 30.01.2021 04:50

Mathematics, 30.01.2021 04:50


Mathematics, 30.01.2021 04:50

Mathematics, 30.01.2021 04:50