
Chemistry, 05.10.2020 21:01 jsotopalomino1016
A diprotic acid, H₂A, has Ka1 = 3.4 × 10⁻⁴ and Ka2 = 6.7 × 10⁻⁹. What is the pH of a 0.18 M solution of H₂A?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:20
Which of these conditions most likely produces an unstable isotope?
Answers: 1

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 09:50
What are four significant sources of ghgs that come from wostem washington?
Answers: 2

Chemistry, 23.06.2019 06:00
What are the coefficients to balance the following equation? ba+br2=babr2
Answers: 2
You know the right answer?
A diprotic acid, H₂A, has Ka1 = 3.4 × 10⁻⁴ and Ka2 = 6.7 × 10⁻⁹. What is the pH of a 0.18 M solution...
Questions






Spanish, 06.07.2019 19:30

Mathematics, 06.07.2019 19:30



Biology, 06.07.2019 19:30

Mathematics, 06.07.2019 19:30



Mathematics, 06.07.2019 19:30

Biology, 06.07.2019 19:30


Biology, 06.07.2019 19:30

Mathematics, 06.07.2019 19:30

History, 06.07.2019 19:30

Mathematics, 06.07.2019 19:30

![[H^+] = (K_{a1} \times C)^{\frac{1}{2}}](/tpl/images/0790/1081/49424.png)
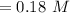
![[H^{+}] = (3.4 \times 10^{-4} \times 0.18 \ M)^{\frac{1}{2}}\\](/tpl/images/0790/1081/9fba4.png)
![[H^{+}] = 0.007\ M](/tpl/images/0790/1081/fc760.png)
![[H^{+}] = K_{a2} \\\\](/tpl/images/0790/1081/99c7c.png)
![[ H^{+} ] = 6.7 \times 10^{-9}\ M \\](/tpl/images/0790/1081/645f7.png)
![Total [H^{+}] = 0.007\ M + 6.7 \times 10^{-9}\ M\\\\Total [H^{+}] = 0.007 \ M](/tpl/images/0790/1081/137ce.png)
![pH = -\log[H^+] \\\\pH = -\log(0.007 \ M)\\\\pH = 2.15](/tpl/images/0790/1081/fd4b3.png)


