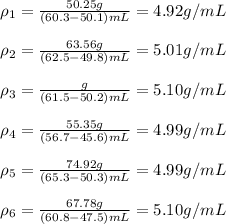Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 10:00
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural.question 2 reflects a moral or social value.question 3 refers to something that can be measured.question 4 reflects a question that can’t be observed.
Answers: 1
You know the right answer?
Claim The student hopes the samples are gold, which has a density of 19.3 g/cm3 . A local geologist...
Questions


Mathematics, 19.09.2019 00:00

Biology, 19.09.2019 00:00



Advanced Placement (AP), 19.09.2019 00:00

Mathematics, 19.09.2019 00:00

Social Studies, 19.09.2019 00:00


Chemistry, 19.09.2019 00:00


Chemistry, 19.09.2019 00:00

Mathematics, 19.09.2019 00:00

Mathematics, 19.09.2019 00:00

History, 19.09.2019 00:00

History, 19.09.2019 00:00

Mathematics, 19.09.2019 00:00