Chemistry, 24.09.2020 01:01 zaratayyibah
Double bonds can act as Lewis bases, sharing their electrons with Lewis acids.
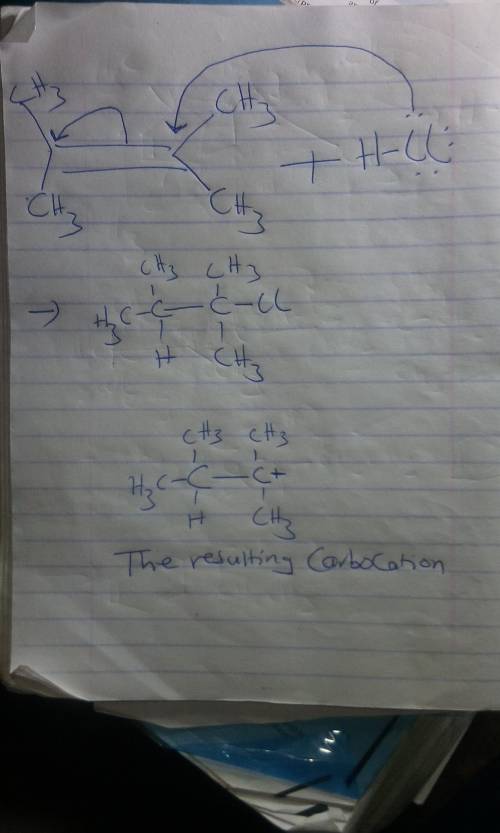
A. Use curved arrows to show how the alkene below will react with HCl.
B. Make the ends of your arrows specify the origin and destination of reorganizing electrons.
C. Draw the resulting carbocation.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2

You know the right answer?
Double bonds can act as Lewis bases, sharing their electrons with Lewis acids.
A. Use curved arrows...
Questions


Physics, 14.10.2020 16:01

Chemistry, 14.10.2020 16:01


Mathematics, 14.10.2020 16:01

English, 14.10.2020 16:01

Biology, 14.10.2020 16:01

Mathematics, 14.10.2020 16:01


Mathematics, 14.10.2020 16:01


Physics, 14.10.2020 16:01

Mathematics, 14.10.2020 16:01


English, 14.10.2020 16:01

Social Studies, 14.10.2020 16:01

Advanced Placement (AP), 14.10.2020 16:01

Biology, 14.10.2020 16:01

Chemistry, 14.10.2020 16:01